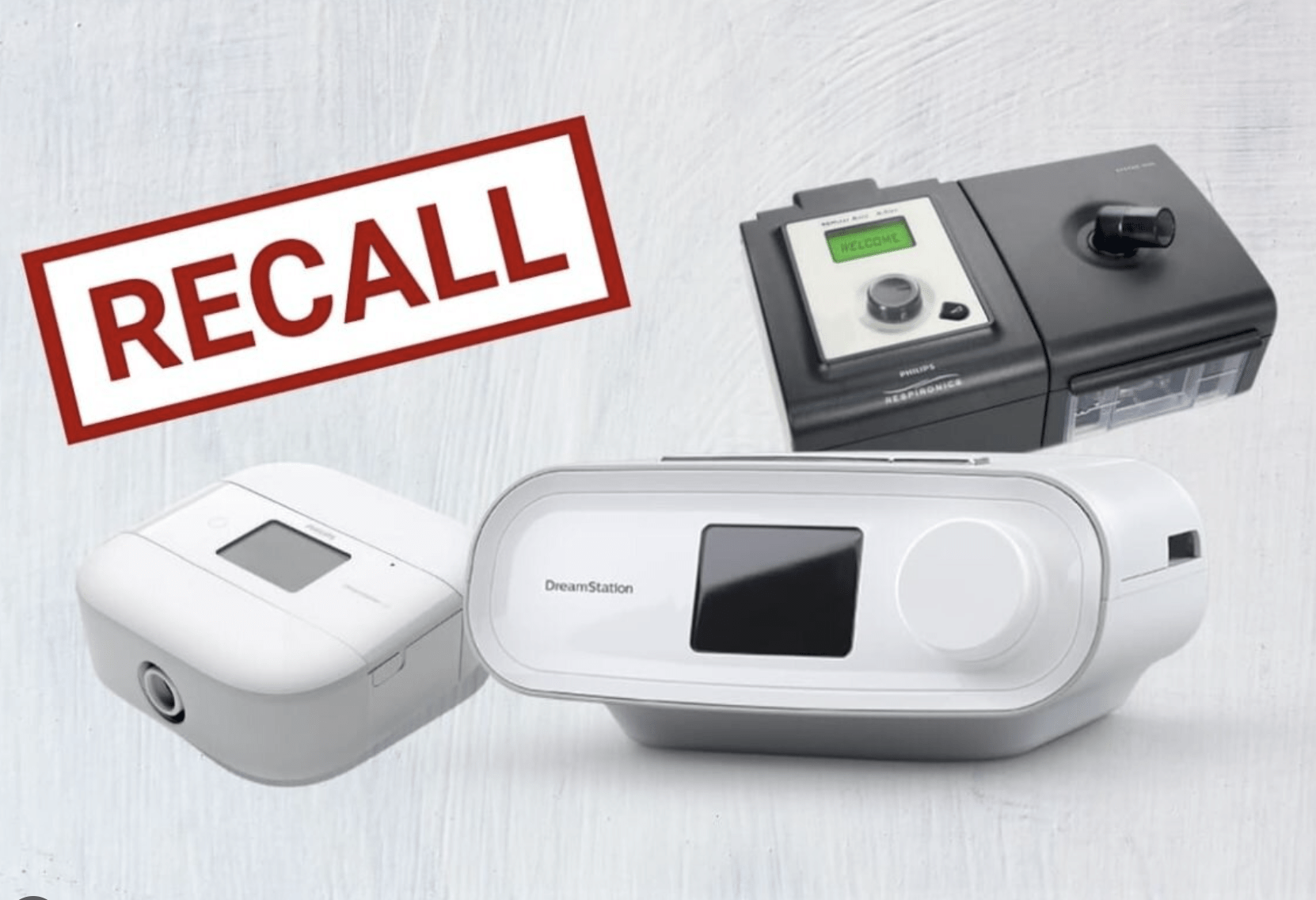
PRO PUBLICA – The first complaints landed at the offices of Philips Respironics in 2010, soon after the company made a fateful decision to redesign its best selling breathing machines used in homes and hospitals around the world.
To silence the irritating rattle that kept users awake at night, Philips packed the devices with an industrial foam — the same kind used in sofas and mattresses. It quickly became clear that something had gone terribly wrong.
The reports coming into Philips described “black particles” or “dirt and dust” inside machines that pump air to those who struggle to breathe. One noted an “oily-like” substance. Others simply warned of “contamination.”
The complaints targeted some of the company’s most celebrated devices built in two factories near Pittsburgh, including ventilators for the sick and dying and the popular DreamStation for patients who suffer from sleep apnea, a chronic disorder that causes breathing to stop and start through the night.
Yet Philips withheld the vast majority of the warnings from the Food and Drug Administration, even as their numbers grew from dozens to hundreds to thousands and became more alarming each year.
“Black shavings in the chamber,” said one 2011 report that was kept from the government. “Contaminated with unknown sticky substance,” noted another three years later.
By 2015, the year Philips launched the DreamStation, the company had amassed at least 25 complaints that pointed to a specific cause — the foam was falling apart.
In June 2021, more than a decade after the first reports, Philips announced a recall of millions of machines that had been delivered to nearly every corner of the United States and dozens of other countries.
The company acknowledged that the foam it had chosen could crumble in heat and humidity and send potentially “toxic and carcinogenic” material into the noses, mouths, throats and lungs of users …